

Use the three sets of mass and volume measurements to calculate three density values for water. Analysis: Subtract the mass of the empty cylinder from each combined mass measurement to obtain three mass measurements of water.Use your thermometer to record the temperature of the water in your graduated cylinder.Repeat Step 3 to obtain a third set of mass and volume measurements.Again, precisely measure this volume of water, and then measure the combined mass using the electronic balance. Add another 20-25 mL of distilled water to the graduated cylinder.Then measure the combined mass using the electronic balance. The exact value is actually slightly lower. The density of water is about 1 gram per milliliter (g/ml), 1 gram per cubic centimeter (g/cm 3 ), 1000 kg/m 3, or 62 pounds per cubic foot (lb/ft 3 ). For all practical purposes, this is the same as the weight of water per unit of volume. Add 20-25 mL of distilled water to the graduated cylinder. The density of water is the mass of water per unit of volume.Using the electronic balance, obtain the mass of your 100-mL graduated cylinder.Once the density is determined in this manner, it will be used to identify the unknown material analyzed. Composite materials such as wood and high-density polyurethane foam contain void spaces which reduce the average density.\] Metals, whose atoms pack together quite compactly, have the highest densities, although that of lithium, the highest metallic element, is quite low. The density range of solids is quite wide. Liquid densities are largely independent of pressure, but they are somewhat temperature-sensitive. Mercury, being a liquid metal, is something of an outlier. Liquids encompass an intermediate range of densities. Measurement of the density of a gas is a simple experimental way of estimating its molecular weight. To the extent that a gas exhibits ideal behavior (low pressure, high temperature), the density of a gas is directly proportional to the masses of its component atoms, and thus to its molecular weight. In general, gases have the lowest densities, but these densities are highly dependent on the pressure and temperature which must always be specified. What we conventionally call the "density" is more precisely known as the "mass density".ĭensity can be expressed in any combination of mass and volume units the most commonly seen units are grams per mL (g mL –1, g cm –3), or kilograms per liter. The general meaning of density is the amount of anything per unit volume. The volume units milliliter (mL) and cubic centimeter (cm 3) are identical and are commonly used interchangeably. This quantity \(\rho\) is known as the density, which is usually defined as the mass per unit volume: Denoting mass and volume by \(m\) and \(V\) respectively, we can write the equation of each line as \(m = \rho V\), where the slope \(\rho\) (Greek lower-case rho) is the proportionality constant that relates mass to volume. The only difference between these plots is their slopes. the plots are all straight lines, which signify direct proportionality.the plots all have the same origin of (0,0): if the mass is zero, so is the volume.These plots show how the masses of three liquids vary with their volumes. The density of seawater is a function of temperature, salinity, and pressure. In oceanography the density of seawater has been expressed historically in grams per cubic centimetre. It is this ratio, (mass ÷ volume), that we are concerned with in this Module. Seawater - Density, Pressure, Salinity: The density of a material is given in units of mass per unit volume and expressed in kilograms per cubic metre in the SI system of units. T he ratio of two extensive properties is always an intensive property - one that characterizes a particular kind of matter, independently of its size or mass. Mass and volume are measures of the quantity of a substance, and as such are defined as extensive properties of matter. For example, according to the table, the density of water at a temperature of 20☌ is 998.2 kg/m 3, and when it is heated to 90☌, the density.
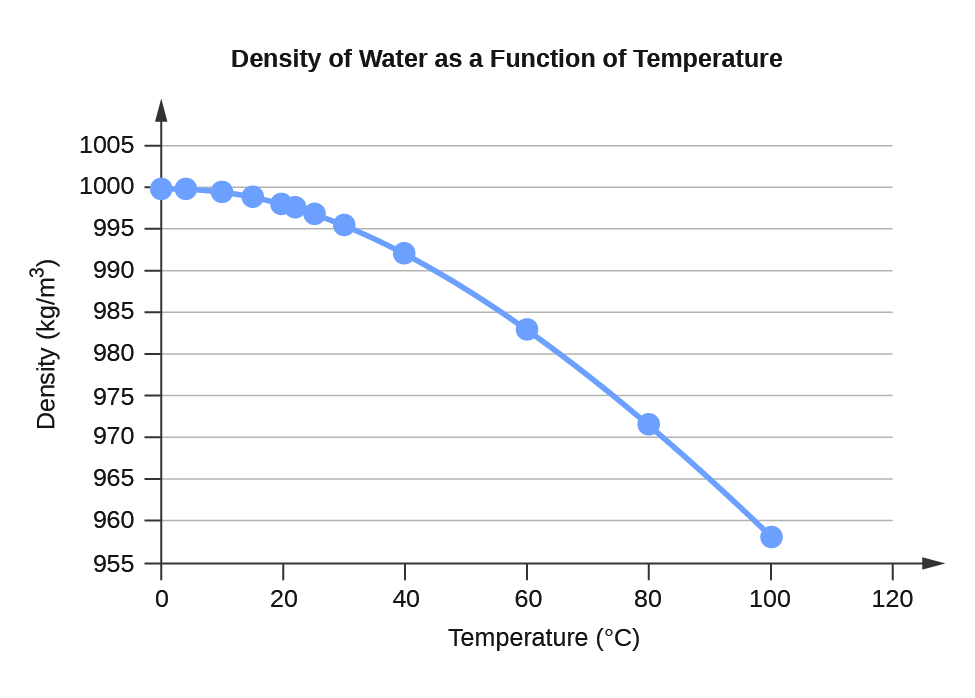
It should be noted that as the water temperature increases (above 4☌), its density decreases. But in making such statements, we are implicitly comparing equal volumes of these substances: after all, we know that a cup of sugar will weigh more than a single ordinary steel nail. The table below shows the density of water in g/ml for different temperatures between 0 and 100 ☌. Most of us have long understood that "oil is lighter than water", or that iron is "heavier" than sugar.
